Home Profile
Profile  Product Range
Product Range Industries
Industries Infrastructure
Infrastructure Our Quality
Our Quality Custom Manufacturing
Custom Manufacturing Network
Network Contact Us
Contact Us Send Enquiry
Send Enquiry



What is Dye
A natural or synthetic substance used to add a color or to change the
color of something. Dyes are the coloring material that color commodities of
our day to day use. Dyes are applied everywhere, from Plastic toys for
children to that fabrics you wear, from food to wood; hardly there is any
industry where dyes are not used commercially.
A dye is a colored substance that has an affinity to the substrate to which
it is being applied. It is an ionising and aromatic organic compounds. The
dye is generally applied in an aqueous solution, and may require a mordant
to improve the fastness of the dye on the fiber.
With the help of dyes we can easily manipulate things according to our
liking. At the very basic level the use of color in identifying individual
components of tissue sections can be accomplished primarily with dyes. Dyes
are applied to numerous substrates for example to textiles, leather,
plastic, paper, food etc. The rule that we apply to other chemicals is
similarly applicable to dyes also. They also get completely or atleast
partially soluble in which it is being put to. For example certain kind of
dyes can be toxic, carcinogenic or mutagenic and can be hazardous to health.
Evolution of Dyes
It was in 2600 BC when earliest written records
of the use of dyestuffs were found in China. The preparation and application
of dyestuffs is one of the oldest forms of human activities. Evidences of
which were found by Excavation at archeological sites where ancient fabrics
were unearthed. There is also mention of it in the Bible and other works of
classical antiquity.
The real breakthroughs in the history of dyes came in 1856 when a teenager
who was experimenting at his makeshift laboratory in home made a certain
discovery that acted as a sort of launching pad for the modern chemicals
industry.
William Perkin an 18-year-old student was working on chemical synthesis of
natural products. In a classic case of serendipity, the young William Perkin
chanced upon his now famous 'Aniline Mauve' dye while he was attempting to
synthesize quinine, the only cure for malaria. Perkin named his color
Mauveine, after the French name of non-fast color which was made of natural
dyes. So "Mauve" (a basic dye) was the first synthetic dye stuff.
Mauve was a derivative of coal tar. It was the first mass-produced dye, that
was commercially available and the idea was born that a color could be made
in the factory. It was indeed a revolution.
What makes the Dyes colored ?
Dyes are basically ionising and aromatic
compounds, they have Chromophores present in them. Their structures have
Aryl rings that has delocalised electron systems. These structures are said
to be responsible for the absorption of electromagnetic radiation that has
varying wavelengths, based upon the energy of the electron clouds.
Chomophores make the dyes proficient in their ability to absorb radiation.
Chromophores act by making energy changes in the delocalised electron cloud
of the dye. This alteration invariably results in the compound absorbing
radiation within the visible range of colors and not outside it. Human eyes
detects this absorption, and responds to the colors.
Electrons may result in loss of color, their removal may cause the rest of
the electrons to revert to the local orbits. A very good example is the
Schiff's reagent. As Sulphurous acid reacts with Pararosanilin, what happens
is that a Sulphonic group attaches itself to the compound's central carbon
atom. This hampers the conjugated double bond system of the Quinoid ring,
and causes the electrons to become localised. As a consequence the ring
ceases to be a Chromophore. As a result, the dye becomes colorless.
To conclude chromophores are the atomic configurations which has
delocalised electrons. Generally they are represented as carbon, nitrogen,
oxygen and sulphur. They can have alternate single and double bonds.
How can the color of the Dyes be altered ?
The color of the dyes are altered by the
Modifiers. The Color modifiers of methyl or ethyl groups are responsible for
any alteration in the dyes; they alter the energy in the delocalised
electrons. There is a progressive alteration of color by adding a particular
modifier. For example: Methyl Violet Series.
The following diagram explains what happens to the color of the dyes
when modifiers are added.
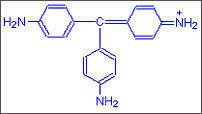 Step
A :
Step
A : When no methylgroup is added the original dye
Pararosanil
as it is called is red in colour.
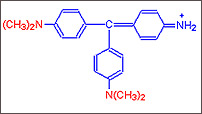 Step
B :
Step
B : As Four Methyl groups are added the reddish purple dye Methyl Violet
is obtained.
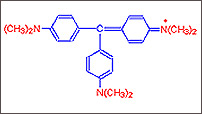 Step
C :
Step
C : With the addition of more groups a purple blue dye Crystal Violet is
obtained. It has in it six such groups.
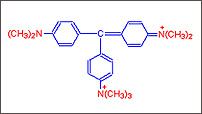 Step
D :
Step
D : Further addition of a seventh methyl group the dye that is obtained
is called
Methyl green.
What gives the Dyes Solubility and Cohesiveness ?
Auxochrome, the only substance responsible for
providing solubility and cohesiveness to dyes. An auxochrome is a group of
atoms attached to a chromophore which modifies the ability of that
chromophore to absorb light. Examples include the hydroxyl group (-OH), the
amino group (-NH2), and an aldehyde group (-CHO).
The presence of an auxochrome in the chromogen molecule is essential to
make a dye. However, if an auxochrome is present in the meta position to the
chromophore, it does not affect the color.
Auxochrome has the ability to intensify colors. It is a group of atoms
which attaches to non-ionising compounds yet has the ability to ionise.
Auxochromes are of two types, positively charged or negatively charged.
Classification of Dyes
Dyes can be classified in several ways, each class
has a very unique chemistry, structure and particular way of bonding. Some
dyes can react chemically with the substrates forming strong bonds in the
process, and others can be held by physical forces. Some of the prominent
ways of classification are given below
- Natural / Synthetic
- Organic / Inorganic
- By area and method of application
- Chemical classification - Based on the nature of their respective
chromophores.
- By nature of the Electronic Excitation (i.e., energy transfer
colorants, absorption colorants and fluorescent colorants).
- According to the dyeing methods
- Anionic (for Protein fibre)
- Direct (Cellulose)
- Disperse (Polyamide fibres)
US International Trade Commission has advocated the most popular
classification of dyes. This system classifies dyes into 12 types, which are
given below:
| Group |
Application |
| Direct |
Cotton, cellulosic and blended fibres |
| Vat dyes |
Cotton, cellulosic and blended fibres |
| Sulphur |
Cotton, cellulosic fibre |
| Organic pigments |
Cotton, cellulosic, blended fabric, paper |
| Reactive |
Cellulosic fibre and fabric |
| Disperse dyes |
Synthetic fibres |
| Acid Dyes |
Wool, silk, paper, synthetic fibres, leather |
| Azoic |
Printing Inks and Pigments |
| Basic |
Silk, wool, cotton |
Home Profile
Profile
 Product
Range
Product
Range Industries
Industries Infrastructure
Infrastructure Our
Quality
Our
Quality Custom
Manufacturing
Custom
Manufacturing Network
Network Contact
Us
Contact
Us Send
Enquiry
Send
Enquiry

![]() Profile
Profile ![]() Product Range
Product Range![]() Industries
Industries![]() Infrastructure
Infrastructure![]() Our Quality
Our Quality![]() Custom Manufacturing
Custom Manufacturing![]() Network
Network![]() Contact Us
Contact Us![]() Send Enquiry
Send Enquiry


 Step
A : When no methylgroup is added the original dye Pararosanil
as it is called is red in colour.
Step
A : When no methylgroup is added the original dye Pararosanil
as it is called is red in colour. Step
B : As Four Methyl groups are added the reddish purple dye Methyl Violet
is obtained.
Step
B : As Four Methyl groups are added the reddish purple dye Methyl Violet
is obtained. Step
C : With the addition of more groups a purple blue dye Crystal Violet is
obtained. It has in it six such groups.
Step
C : With the addition of more groups a purple blue dye Crystal Violet is
obtained. It has in it six such groups. Step
D : Further addition of a seventh methyl group the dye that is obtained
is called Methyl green.
Step
D : Further addition of a seventh methyl group the dye that is obtained
is called Methyl green.![]() Profile
Profile
![]() Product
Range
Product
Range![]() Industries
Industries![]() Infrastructure
Infrastructure![]() Our
Quality
Our
Quality![]() Custom
Manufacturing
Custom
Manufacturing![]() Network
Network![]() Contact
Us
Contact
Us![]() Send
Enquiry
Send
Enquiry